This means that atomic mass units can be easily converted to kilograms or grams. For example, carbon-12 has a mass of 12 amu. By multiplying 12 amu by 1.66 x 10^-27 kg/amu we find the mass of. An amu is a unit of mass, like a pound, gram, microgram, or tonne. An amu is a very, very small unit of mass, defined, as you know by now, as being exactly the mass of 12 isolated 12 C atoms in their ground state (which is just a way to eliminate kinetic or potential energy which would give the atoms more mass (remember E = mc²)).
Atomic Mass To Grams
Please provide values below to convert dalton to gram [g], or vice versa.
Amu To Grams Formula
Dalton to Gram Conversion Table
| Dalton | Gram [g] |
|---|---|
| 0.01 dalton | 1.6605300000013E-26 g |
| 0.1 dalton | 1.6605300000013E-25 g |
| 1 dalton | 1.6605300000013E-24 g |
| 2 dalton | 3.3210600000027E-24 g |
| 3 dalton | 4.981590000004E-24 g |
| 5 dalton | 8.3026500000067E-24 g |
| 10 dalton | 1.6605300000013E-23 g |
| 20 dalton | 3.3210600000027E-23 g |
| 50 dalton | 8.3026500000067E-23 g |
| 100 dalton | 1.6605300000013E-22 g |
| 1000 dalton | 1.6605300000013E-21 g |
How to Convert Dalton to Gram
1 dalton = 1.6605300000013E-24 g
1 g = 6.02217364335E+23 dalton
Example: convert 15 dalton to g:
15 dalton = 15 × 1.6605300000013E-24 g = 2.490795000002E-23 g
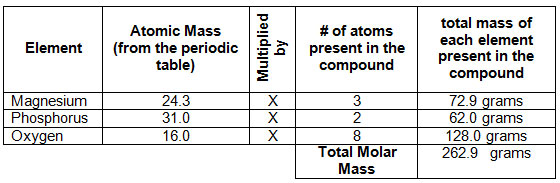
How To Convert Amu To Gram

Popular Weight And Mass Unit Conversions

Convert Dalton to Other Weight and Mass Units
Amu To Grams Formula
What is the mathematical relationship between atomic mass units (amu) and grams (g)?
1 gram = ? amu
1g of H atoms = 1 mole of H atoms = 6.02 x 10^23 atoms. The atomic mass of H = 1 amu, so you could work out the mass of 1 x H atom in grams as 1/6.02 x 10^23 g = 1.66 x 10^-24g.
4g of He atoms = 1 mole of He atoms = 6.02 x 10^23 atoms. The atomic mass of He = 4 amu, so you could work out the mass of 1 x He atom in grams as 4/6.02 x 10^23 g = 6.64 x 10^-24g (4 times the mass of the each H atom) .
Amu To Grams Problems
So you could argue that each amu has a mass of 1.66 x 10^-24g. However the gram scale is an actual mass scale whereas the amu scale is a relative scale based on C-12 having a mass of 12 amu,.
